Authors
Hou, Jing, Chen, Kai-Xuan, He, Chen, Li, Xiao-Xiao, Huang, Mei, Jiang, Yang-Zi, Jiao, Yu-Rui, Xiao, Qiao-Ni, He, Wen-Zhen, Liu, Ling, Zou, Nan-Yu, Huang, Min, Wei, Jie, Xiao, Ye, Yang, Mi, Luo, Xiang-Hang, Zeng, Chao, Lei, Guang-Hua, Li, Chang-Jun
Lab
Journal
Nature Aging
Abstract
The accumulation and systemic propagation of senescent cells contributes to physiological aging and age-related pathology. However, which cell types are most susceptible to the aged milieu and could be responsible for the propagation of senescence has remained unclear. Here we found that physiologically aged bone marrow monocytes/macrophages (BMMs) propagate senescence to multiple tissues, through extracellular vesicles (EVs), and drive age-associated dysfunction in mice. We identified peroxisome proliferator-activated receptorAlpha (PPARAlpha) as a target of microRNAs within aged BMM-EVs that regulates downstream effects on senescence and age-related dysfunction. Demonstrating therapeutic potential, we report that treatment with the PPARAlpha agonist fenofibrate effectively restores tissue homeostasis in aged mice. Suggesting conservation to humans, in a cohort study of 7,986 participants, we found that fenofibrate use is associated with a reduced risk of age-related chronic disease and higher life expectancy. Together, our findings establish that BMMs can propagate senescence to distant tissues and cause age-related dysfunction, and they provide supportive evidence for fenofibrate to extend healthy lifespan. Hou, Chen et al. show that aged bone marrow macrophages propagate senescence to multiple tissues in vivo, through extracellular vesicles containing PPARAlpha-targeted microRNAs. They demonstrate the therapeutic potential of intervening in this process using the PPARAlpha agonist fenofibrate.
Keywords/Topics
marrow;macrophages;drive;systemic;aging;age-related;dysfunction;extracellular;vesicle-mediated;induction
BIOSEB Instruments Used:
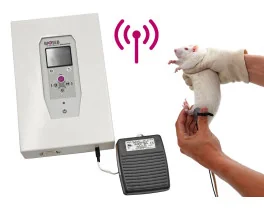
Grip strength test (BIO-GS4)
Source :

 Douleur - Allodynie/Hyperalgésie Thermique
Douleur - Allodynie/Hyperalgésie Thermique Douleur - Spontanée - Déficit de Posture
Douleur - Spontanée - Déficit de Posture Douleur - Allodynie/Hyperalgésie Mécanique
Douleur - Allodynie/Hyperalgésie Mécanique Apprentissage/Mémoire - Attention - Addiction
Apprentissage/Mémoire - Attention - Addiction Physiologie & Recherche Respiratoire
Physiologie & Recherche Respiratoire





























 Douleur
Douleur Système Nerveux Central (SNC)
Système Nerveux Central (SNC)  Neurodégénérescence
Neurodégénérescence Système sensoriel
Système sensoriel Système moteur
Système moteur Troubles de l'humeur
Troubles de l'humeur Autres pathologies
Autres pathologies Système musculaire
Système musculaire Articulations
Articulations Métabolisme
Métabolisme Thématiques transversales
Thématiques transversales Congrès & Meetings 2026
Congrès & Meetings 2026 