Authors
Daniel C. Andersson, Matthew J. Betzenhauser, Steven Reiken, Alisa Umanskaya, Takayuki Shiomi, Andrew R. Marks
Lab
College of Physicians and Surgeons of Columbia University, New York, USA
Journal
The Journal of Physiology
Abstract
Enhancement of contractile force (inotropy) occurs in skeletal muscle following neuro-endocrine release of catecholamines and activation of muscle ?-adrenergic receptors. Despite extensive study, the molecular mechanism underlying the inotropic response in skeletal muscle is not well understood. Here we show that phosphorylation of a single serine residue (S2844) in the sarcoplasmic reticulum (SR) Ca2+ release channel/ryanodine receptor type 1 (RyR1) by cAMP-dependent protein kinase A (PKA) is critical for skeletal muscle inotropy. Treating fast-twitch skeletal muscle from wild type mice with the ?-receptor agonist isoproterenol (ISO) increased RyR1 PKA phosphorylation, twitch Ca2+ and force generation. In contrast, the enhanced muscle Ca2+, force and in vivo muscle strength responses following ISO stimulation were abrogated in RyR1-S2844A mice in which the Serine in the PKA site in RyR1 was replaced with alanine. These data suggest that the molecular mechanism underlying skeletal muscle inotropy requires enhanced SR Ca2+ release due to PKA phosphorylation of Serine 2844 in RyR1.
BIOSEB Instruments Used
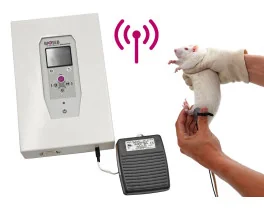
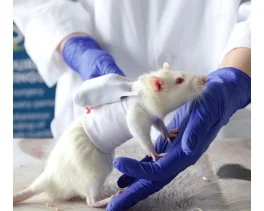
Grip strength test (BIO-GS3)
Keywords/Topics
Stress; Fonctions squelettales; Troubles de l'humeur; Système musculaire
Source :
http://onlinelibrary.wiley.com/doi/10.1113/jphysiol.2012.237925/abstract

 Douleur - Allodynie/Hyperalgésie Thermique
Douleur - Allodynie/Hyperalgésie Thermique Douleur - Spontanée - Déficit de Posture
Douleur - Spontanée - Déficit de Posture Douleur - Allodynie/Hyperalgésie Mécanique
Douleur - Allodynie/Hyperalgésie Mécanique Apprentissage/Mémoire - Attention - Addiction
Apprentissage/Mémoire - Attention - Addiction Physiologie & Recherche Respiratoire
Physiologie & Recherche Respiratoire





























 Douleur
Douleur Système Nerveux Central (SNC)
Système Nerveux Central (SNC)  Neurodégénérescence
Neurodégénérescence Système sensoriel
Système sensoriel Système moteur
Système moteur Troubles de l'humeur
Troubles de l'humeur Autres pathologies
Autres pathologies Système musculaire
Système musculaire Articulations
Articulations Métabolisme
Métabolisme Thématiques transversales
Thématiques transversales Congrès & Meetings
Congrès & Meetings 