Authors
A. Meyer, G. Laverny, Y. Allenbach, E. Grelet, V. Ueberschlag, A. Echaniz-Laguna, B. Lannes, G. Alsaleh, A. Charles, F. Singh, J. Zoll, E. Lonsdorfer, F. Maurier, O. Boyer, J. E. Gottenberg, A. S. Nicot, J. Laporte, O. Benveniste, D. Metzger, J. Sibilia, B. Geny Gottenberg JE12,4, Nicot AS4,5, Laporte J4,5, Benveniste O6, Metzger D4,5, Sibilia J12,4, Geny B9,4. Lannes B8, Alsaleh G4, Charles AL9,4, Singh F9,4, Zoll J9,4, Lonsdorfer E9,4, Maurier F10, Boyer O11, Gottenberg JE1
Lab
Hôpitaux Universitaires de Strasbourg, Institut de Physiologie, Strasbourg, France
Journal
Acta Neuropathologica: Pathology and Mechanisms of Neurological Disease
Abstract
Dermatomyositis (DM) is an autoimmune disease associated with enhanced type I interferon (IFN) signalling in skeletal muscle, but the mechanisms underlying muscle dysfunction and inflammation perpetuation remain unknown. Transcriptomic analysis of early untreated DM muscles revealed that the main cluster of down-regulated genes was mitochondria-related. Histochemical, electron microscopy, and in situ oxygraphy analysis showed mitochondrial abnormalities, including increased reactive oxygen species (ROS) production and decreased respiration, which was correlated with low exercise capacities and a type I IFN signature. Moreover, IFN-β induced ROS production in human myotubes was found to contribute to mitochondrial malfunctions. Importantly, the ROS scavenger N-acetyl cysteine (NAC) prevented mitochondrial dysfunctions, type I IFN-stimulated transcript levels, inflammatory cell infiltrate, and muscle weakness in an experimental autoimmune myositis mouse model. Thus, these data highlight a central role of mitochondria and ROS in DM. Mitochondrial dysfunctions, mediated by IFN-β induced-ROS, contribute to poor exercise capacity. In addition, mitochondrial dysfunctions increase ROS production that drive type I IFN-inducible gene expression and muscle inflammation, and may thus self-sustain the disease. Given that current DM treatments only induce partial recovery and expose to serious adverse events (including muscular toxicity), protecting mitochondria from dysfunctions may open new therapeutic avenues for DM.
BIOSEB Instruments Used
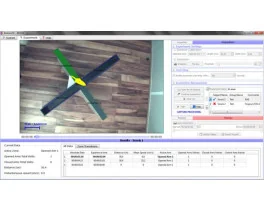
Grip strength test (BIO-GS3)
Keywords/Topics
Motor Deficits; Skeletal functions; Motor control; Muscular system; Autoimmune disease; Exercise & Endurance
Source :

 Pain - Thermal Allodynia / Hyperalgesia
Pain - Thermal Allodynia / Hyperalgesia Pain - Spontaneous Pain - Postural Deficit
Pain - Spontaneous Pain - Postural Deficit Pain - Mechanical Allodynia / Hyperalgesia
Pain - Mechanical Allodynia / Hyperalgesia Learning/Memory - Attention - Addiction
Learning/Memory - Attention - Addiction Physiology & Respiratory Research
Physiology & Respiratory Research










![Dynamic Weight Bearing 2.0 – Postural Module [Add-on]](https://bioseb.com/733-home_default/dynamic-weight-bearing-20-add-on-postural-module.jpg)



















 Pain
Pain Central Nervous System (CNS)
Central Nervous System (CNS) Neurodegeneration
Neurodegeneration Sensory system
Sensory system Motor control
Motor control Mood Disorders
Mood Disorders Other disorders
Other disorders Muscular system
Muscular system Joints
Joints Metabolism
Metabolism Cross-disciplinary subjects
Cross-disciplinary subjects CONFERENCES & MEETINGS 2026
CONFERENCES & MEETINGS 2026 